 |
| Molecular Expressions Center: FSU |
An interesting study was published in this month’s issue of
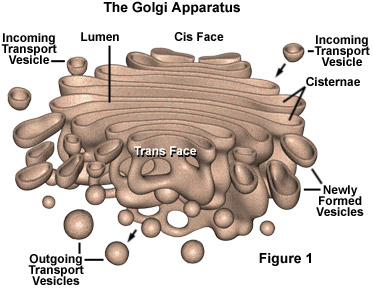
Cell exploring the response of the Golgi Apparatus to DNA damage in cells. The
Golgi typically functions to package proteins immediately after synthesis in
the nucleus, preparing them for transport to their various destinations in the
cell or secretion out of it. While the response of the nucleus to DNA damage is
fairly well understood, less has been done to investigate the effects on other
parts of the cell. This research group, has discovered that, after treatment with DNA damaging
drugs, the Golgi apparatus of all the mammalian cells in the study would break
up and disperse away from the nucleus. My first thought was that this dispersal
was in some way related to the pre-programmed cell death (apoptosis) that
typically begins after DNA damage, but subsequent tests have shown that this
effect was seen in cells that hadn’t begun to apoptose and, moreover, that the
breakdown was quite different to what is seen in cells that are dying. The
three compartments of the Golgi stay associated and organized while the overall structure
disperses, and cells with the Golgi taken apart remain so for weeks after the
cell has recovered from the damage and begun to grow.
The investigators also identify a mechanism that triggers
this dispersal. A protein that is turned on in response to DNA damage, DNA-PK,
activates a Golgi protein known as GOLPH3, and that this activation is
required for the eventual dispersal of the Golgi. This group of investigators
had previously shown that GOLPH3 connects the golgi to the actin cyctoskeleton, a network of tubes that is responsible for physically maneuvering things inside the cell itself.
Further experiments in this paper demonstrated that GOLPH3 has another
important function: helping the cell to resist the pre-programmed cell death
signal in response to DNA damage.
This type of damage activates a very complex balancing act
for damaged cells. While most mutations caused by DNA damage are benign or will
simply lead to the cell dying, some have the potential to lead to cancer. This
leaves an evolutionary problem for damaged cells: if cells die every time they
experience DNA damage, organisms will be constantly forced to replace these,
incurring a massive energy cost. On the other hand, if cells are too tolerant
of DNA damage, the chance of cancer in the afflicted organisms will be much higher. Thus,
selection has led to two very intricate cellular systems opposing each other in
damaged cells: senescence (essentially the cell pausing to repair the damage
before resuming growth) and apoptosis. GOLPH3 serving as an anti-apoptosis
factor makes some sense, as dispersal of the Golgi would inhibit gene
expression in damaged cells, giving them more time for repairs and preventing
the damage from inducing cancer. This is supported by the fact that a number of
cancerous cell lines over express GOLPH3. This does, however, open up an
interesting question with regard to cancer drugs. While drugs that target
GOLPH3 may now be investigated for use in chemotherapy, the fact that it
induces an anti-apoptotic effect in cells with DNA damage have interesting
consequences for a number of other cancer drugs. Many of these drugs induce DNA
damage themselves, causing rapidly growing cells to die in response, but
potentially also inducing activation of GOLPH3 to inhibit their effect. This
will be an interesting area of research to keep an eye on going forward, as it
may explain one of the mechanisms causing cancer drugs to be effective at
treating some patients but ineffective in others.
Comments
Post a Comment